Fig. 1: Formation of diimine by the reaction between phenylenediamine and isobutyraldehyde in acetonitrile.
Imines, also named Schiff’s bases, are easy to synthesize and allow the use of a large variety of starting materials (i.e. amine and aldehyde) for the condensation reaction. Therefore, these compounds are very popular in different areas. In coordination chemistry, Schiff’s bases are used as ligands to obtain metal complexes, like the Salen ligand or the ligand of the Jacobsen´s catalyst. In the dyes and pigments area, metal complex dyes of nickel with Schiff’s bases are used. On the other hand, porphyrin-Schiff’s base ligand compounds have photoluminescence and/or electrochemical activity. In pharmacy, numerous Schiff’s bases are potential bioactive cores, that can have useful biological activities. In biochemistry, Schiff’s bases are commonly used as enzymatic intermediates. Therefore, online monitoring of this reaction is important to understand the mechanism and the formation of intermediates. In this blog post, the application of benchtop NMR to study this kind of reaction is demonstrated taking as an example the reaction between phenylenediame and isobutyraldehyde in acetonitrile to form the diimine product (see Fig. 1). The results reported in this note are in good agreement with data collected on a 400 MHz spectrometer previously reported in reference.
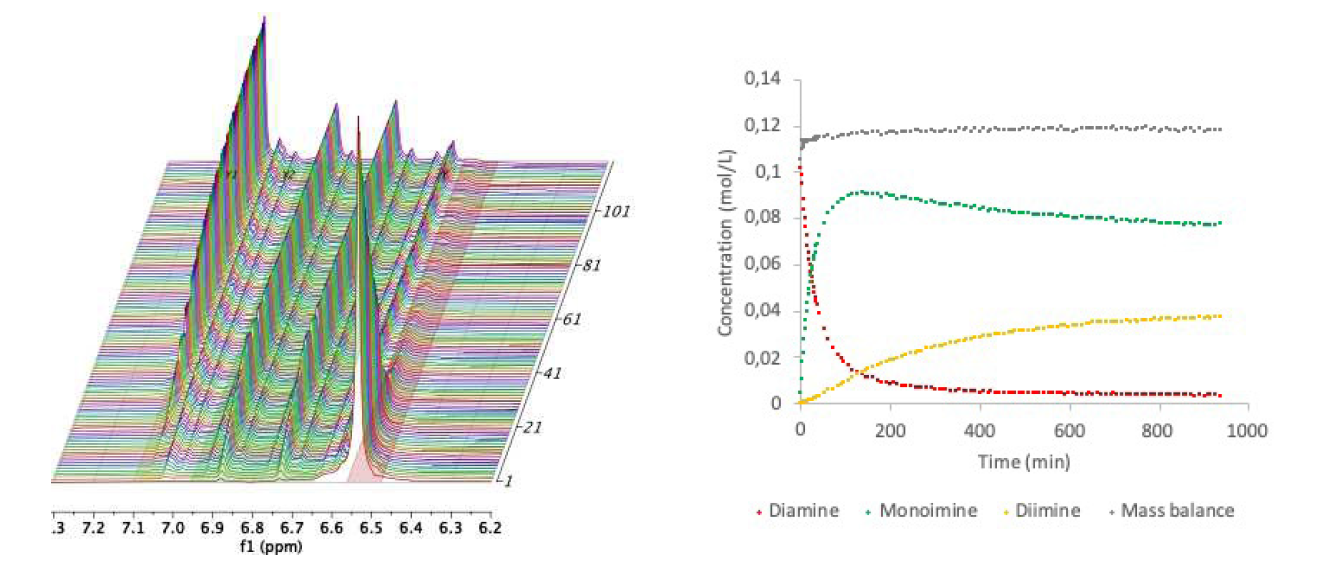
Figure 2 shows a zoom of the spectra collected during the reaction where the aromatic region can be observed. By integrating the signals corresponding to the different products we can see that the concentration of phenylenediamine (6.54 ppm) decreases with time, while the concentrations of mono- and diimine (at 6.80 ppm and 7.06, respectively) increase, showing the typical behaviours of a two step reaction. After approximatively 2 hours, the concentration of monoimine starts to decrease as phenylenediamine is almost completely consumed and the monoimine gets converted into diimine. After a few hours, it can be observed that almost all the starting materials disappeared (96.1%), but still the concentration of monoimine remains relatively high. This is the behaviourr expected in a situation where the kinetic constant of the first step is much larger than the kinetic constant of the second step.
Fig. 2: Left: Zoom of the aromatic region showing the decrease of phenylene diamine (red) and the growth of mono- and diimine (green and yellow), respectively. Right: Integrals of the different signals plotted as a function of time converted to concentration of phenylene diamine (red), monoimine (green) and diimine (yellow).