Reaction Monitoring
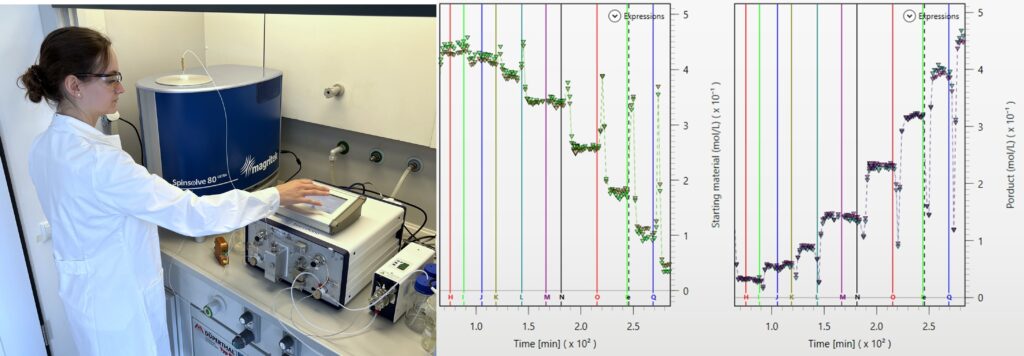
The Spinsolve high-resolution benchtop NMR spectrometer can be installed directly in the fume hood of a chemistry lab to monitor the progress of chemical reactions on-line.
Reaction Monitoring
The Spinsolve high-resolution benchtop NMR spectrometer can be installed directly in the fume hood of a chemistry lab to monitor the progress of chemical reactions on-line.
NMR reaction monitoring is an excellent technique for determining reaction kinetics as well as reaction end points. NMR measurements are quantitative and NMR signals change in a linear way as concentrations vary. The NMR technique is not sensitive to the matrix of the sample. NMR measurements are non-destructive and the complete sample material being inside the NMR coil is analyzed. The Spinsolve high-resolution benchtop NMR spectrometer can be installed directly in the fume hood of a chemistry lab to monitor the progress of chemical reactions on-line. Reactants can be pumped in continuous mode from the reactor to the magnet and back using just standard PTFE tubing or using a glass flow cell. Magritek has created two solution kits for reaction monitoring: one using a glass flow tube with an expanded 4 mm ID section in the measurement zone. The other one, is a very cost-effective solution using just PTFE tubing and a glass guide tube, as well as the pump (read more…). Besides the hardware, Magritek developed a reaction monitoring software module for an easy set-up of the reaction loops and the data processing (read more…).
NMR is an invaluable tool for real-time monitoring of chemical reactions, with capabilities including but not limited to the measurement of percent conversion, kinetic studies, side product formation, and global reaction optimization1,2,3,4. Driven by a collaborative project between Magritek and process chemists from major pharmaceutical companies, as a part of an Emerging Technologies Consortium project on the development of a Portable NMR Reaction Monitoring Platform, we have developed the new Spinsolve Sample Temperature Control system (STC) which allows for online measurements of the reaction mixture from room temperature up to 60 °C.
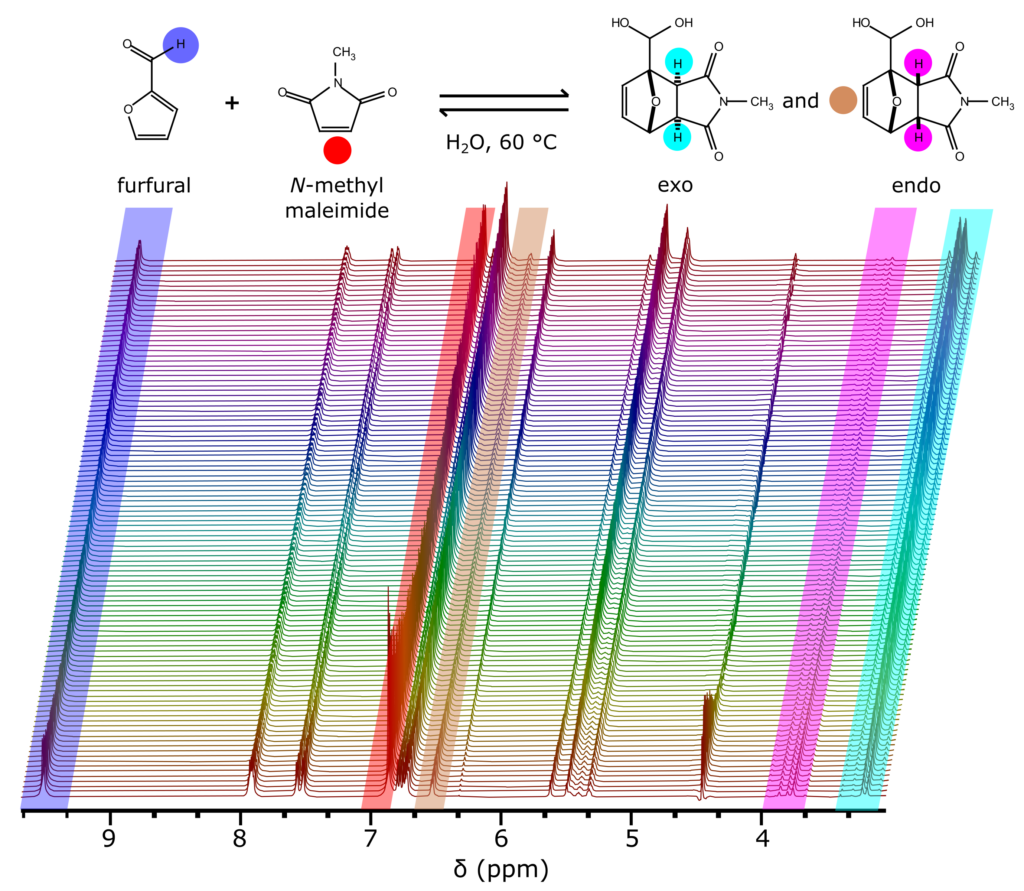
Industrial chemical processes are commonly run at elevated temperature due to solubility or reactivity considerations; therefore, room temperature NMR measurements are applicable to a limited reaction scope. To demonstrate the necessity of sample temperature control technology to study certain reactions with benchtop NMR, in this blog post we monitor, in an NMR tube, a Diels-Alder reaction (DA) between N-methyl maleimide and furfural based on the approach of Cioc R. et al 5 (see the reaction scheme in the picture below). We show that the reagents are insoluble at ambient temperature, while at 60 °C the mixture becomes homogenous. Read more …
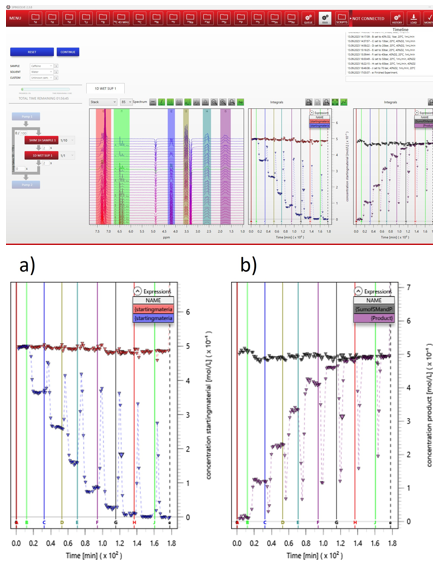
In this application note, we leverage the flexibility, compactness, and robustness of benchtop NMR spectroscopy to present the first experimental setup that enables simultaneous on-line monitoring of both phases of a biphasic reaction system within a single experiment. By integrating phase-selective extraction with two independent benchtop NMR spectrometers, both solvent layers can be continuously analyzed in real time, eliminating the need for repeated experiments and enabling direct comparison of concentration profiles across phases.
The capabilities of this setup are demonstrated using a Suzuki–Miyaura coupling reaction performed under biphasic conditions. For the first time, benchtop NMR spectroscopy is used to non-destructively and quantitatively monitor the reaction kinetics in both phases simultaneously. This approach provides detailed insight into the formation and consumption of reactants, intermediates, side products, and the desired coupling product, while also revealing the time-dependent distribution of chemical species between the aqueous and organic phases. Such information is essential for understanding interphase mass transfer, catalyst speciation, and selectivity-determining steps in biphasic catalysis.
These results are enabled by the high stability, sensitivity, and excellent magnetic field homogeneity of the Spinsolve ULTRA spectrometer, which allows accurate quantification directly in conventional (non-deuterated) solvents. Moreover, the fully in-line configuration permits operation under inert conditions, minimizing oxygen exposure associated with manual sampling and preserving sensitive catalytic systems. Read more …
Recent advances in laboratory automation and analytical instrumentation have opened new possibilities for accelerating reaction development, particularly in flow chemistry. Among these, the integration of real-time analytics with intelligent process control has enabled the emergence of self-optimizing reactor systems. These setups are capable of autonomously exploring reaction conditions to maximize the performance of the reactor. The core to implement self-optimization methods requires an optimization algorithm coupled to an analytical tool that monitors the reaction mixture in real time. In this application note, we present a self-optimizing flow reactor system that integrates the Spinsolve Ultra benchtop NMR with the LabManager® and LabVision® automation tools from HiTec Zang and an Ehrfeld Micro Reaction System. Benchtop NMR spectroscopy has proven to be a powerful online analytical tool that can provide real-time insight into reaction progress. This system was used to continuously monitor and adjust the reaction conditions of a Knoevenagel-Condensation to identify optimal parameters with minimal human intervention. While the continuous microreactor setup used here offers enhanced control, faster reaction dynamics, and seamless integration of feedback for real-time optimization, it is worth mentioning that the automation system is suited as well for batch reactor setups. Read more …
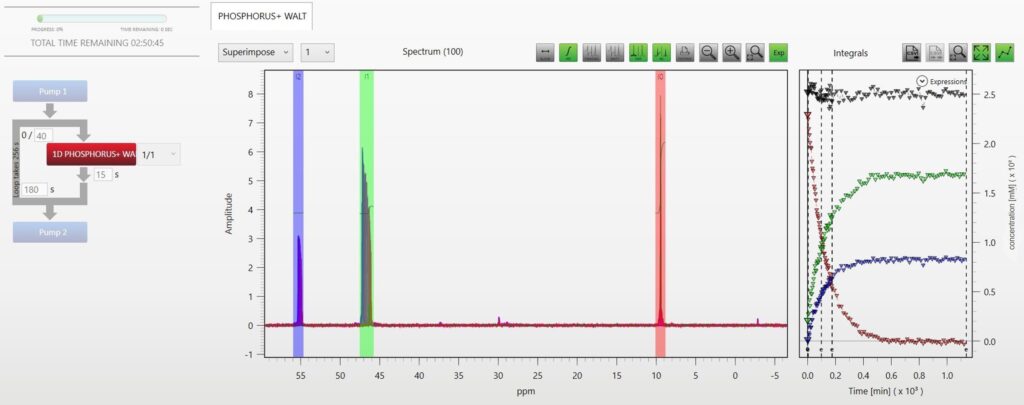
In this Application note we explore the performance of the Spinsolve Benchtop NMR spectrometers for the identification of phosphine ligands by 31P NMR. Moreover, we demonstrate that the sensitivity of the Spinsolve spectrometers is high enough to monitor the oxidation reaction of these ligands in real time. Phosphines and related phosphorus-containing compounds have been extensively used over the past decades for homogeneous metal complex catalysis. This type of catalysis was introduced in the early 1960s. Initially, more active transition metals like palladium and rhodium were employed. In addition, the metal cobalt was as well often modified by phosphine ligands.[1] Since then, a large number of articles have been published reporting on the electronic[2] and steric[3] properties of phosphine ligands and their effects on catalytic reactions. As these phosphine ligands are very often air sensitive, NMR has proven to be the method of choice for their characterization, simply because the NMR analysis is non-invasive and the sample can be prepared in sample tubes with an inert atmosphere. Read more …
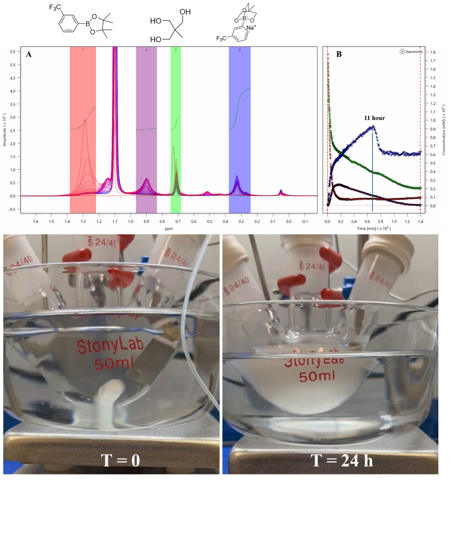
A growing application for Spinsolve benchtop NMR spectrometers is reaction monitoring, where chemists can follow their reactions on-line and get real-time feedback about the reaction progress, observe short-lived intermediates, or detect other physical and chemical changes in the reaction mixture. The implementation of closed-loop sample transfer systems to follow chemical reactions with homogenous liquid mixtures has been described in previous Magritek applications notes (Quantifying the Formation of Stereoisomers, Optimising a Two-Step Hydrogenation Reaction) and numerous journal papers. However, depending on the chemistry, the reaction mixture can change from homogeneous (all the species are in solution) to heterogeneous (presence of precipitation, or immiscible liquids). Monitoring heterogeneous mixtures with NMR is challenging due to the presence of solids or multiple phases in the samples, resulting in loss of spectral resolution (due to line broadening) as well as possible clogging of the transfer lines, which can hinder the sampling process and even cause safety issues. Read more …
In previous appnotes, we have reported on the potential of using a Spinsolve benchtop NMR spectrometer to monitor on-line different hydrogenations conducted on an H-Cube® Pro flow reactor. here. As a follow-up, in this application note, we demonstrate how straightforward it is to perform deuteration reactions employing the H-Cube Pro® flow reactor. This can be achieved by simply replacing the water used to produce H2 (electrolysis) with heavy water. In this application note, we connected a Spinsolve 80 MHz ULTRA benchtop NMR spectrometer from Magritek at the outlet of an H-Cube® Pro flow reactor from ThalesNano©. The Spinsolve ULTRA model allowed us to capture high-resolution spectra of the different products even though they are dissolved in protonated solvents. The H-Cube® Pro offers the flexibility to optimize a reaction in flow, allowing adjustments to temperature, pressure, and hydrogen amount, while also efficiently reducing catalyst loading. In our previous study, we optimized the reaction conditions for the hydrogenation of the double bond of cinnamyl alcohol. In this work, we replaced the water within the tank with heavy water. Consequently, we aimed to illustrate that an optimization of the deuteration of the double bond can be simply achieved through the combination of a Spinsolve benchtop NMR spectrometer
and an H-Cube® Pro. Read more …
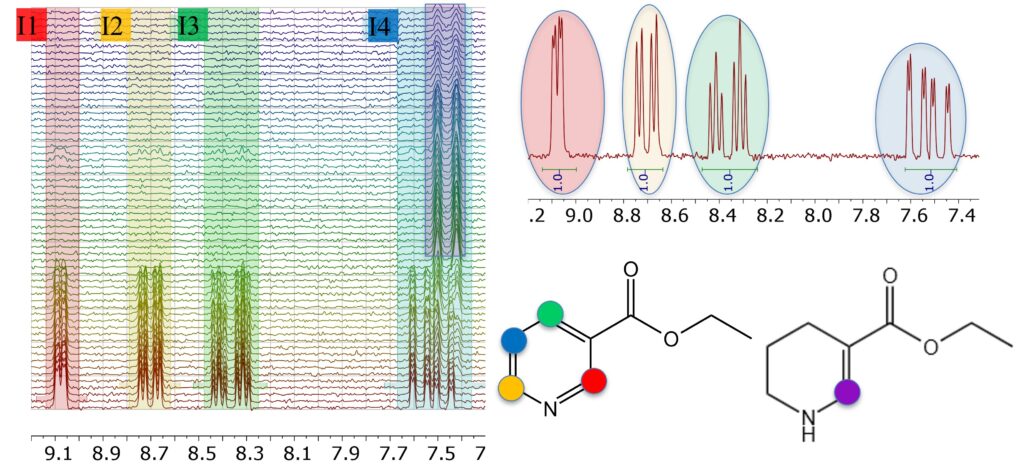
In a recent appnote we demonstrated that coupling a Spinsolve benchtop NMR spectrometer to a continuous flow reactor can dramatically accelerate the optimization of the reaction parameters. Benchtop NMR spectroscopy provides online access to quantify the reaction conversion in real time as the reaction parameters are adjusted. The example chosen in the previous work to test the setup and the performance of benchtop NMR was the hydrogenation reaction of the double bond in cinnamyl alcohol. In that case, the quantification of starting material and product was straightforward. The simplicity of the NMR spectra of those compounds was useful to evaluate the reproducibility and robustness of the NMR method. In this application note, we explore the power of this setup to optimize the more complex two-step hydrogenation reaction of the hetero-aromatic ring of ethyl nicotinate (see Scheme 1) as a function of temperature, pressure, equivalents of H2, and flow rate. Read more …
The synergy between continuous flow reactors and benchtop Nuclear Magnetic Resonance (NMR) spectroscopy has attracted significant attention recently due to its transformative impact on chemical synthesis and analysis. Continuous flow reactors offer distinct advantages, including precise control over reaction parameters, enhanced safety, and improved reaction efficiency. Conversely, benchtop NMR spectroscopy provides, non-destructive, and quantitative insights into reaction kinetics, intermediates, and final products. This integration enables real-time monitoring of the conversion as a function of the reactor parameters, facilitating rapid and even automated reaction optimization. The benefits of this hybrid approach are evident in the accelerated processes of reaction discovery and optimization. Researchers can utilize real-time NMR data to make on-the-fly adjustments to reaction conditions, resulting in enhanced yields and selectivity. Furthermore, in-line analysis reduces the need for laborious sample preparation and off-line analysis, thus streamlining workflows and minimizing waste. Read more …
Imines, also named Schiff’s bases, are easy to synthesize and allow the use of a large variety of starting materials (i.e. amine and aldehyde) for the condensation reaction. Therefore, these compounds are very popular in different areas. In coordination chemistry, Schiff’s bases are used as ligands to obtain metal complexes, like the Salen ligand or the ligand of the Jacobsen´s catalyst. In the dyes and pigments area, metal complex dyes of nickel with Schiff’s bases are used. On the other hand, porphyrin Schiff’s base ligand compounds have photoluminescence and/or electrochemical activity. In pharmacy, numerous Schiff’s bases are potential bioactive cores, that can have useful biological activities. In biochemistry, Schiff’s bases are commonly used as enzymatic intermediates. Therefore, online monitoring of this reaction is important to understand the mechanism and the formation of intermediates. In this note, the application of benchtop NMR to study this kind of reaction is demonstrated taking as an example the reaction between phenylenediame and isobutyraldehyde in acetonitrile to form the diimine product. Read more …
Figure. Left: Formation of diimine by the reaction between phenylenediamine and isobutyraldehyde in acetonitrile. Right: Stacked plot of 1D 1H NMR data of the aromatic region showing the decrease of phenylene diamine (red) and the growth of mono- and diimine (green and yellow), respectively. Integrals of the different signals plotted as a function of time converted to concentration of phenylene diamine (red), monoimine (green) and diimine (yellow).
On-line monitoring of chemical reactions is a fast-growing field finding applications in R&D labs, pilot plants, and large chemical production plants where fully automated analyzers provide feedback to control the reactor. With the availability of the Spinsolve compact benchtop NMR systems, the NMR spectrometer can be brought directly next to the reaction set-up in the lab or even in plant. Permanent magnet systems do not need cryogens or special maintenance and only minimal training is required for personnel to perform the measurements due to the ease of use. In this application note we show the performance of a Spinsolve 60 MHz ULTRA to monitor the N-Acetylation of L-Phenylalanine with acetic anhydride. L-Phenylalanine is an essential amino acid used in a variety of industrial processes, e.g. as reactant in the synthesis of the ACE-inhibitor Alacepril, as well as in the production of the well-known, non-saccharide sweetener Aspartam. In many chemical processes involving amino acids the reactive centers must be protected to ensure the required regioselectivity of the reaction. In this example the acetylation of the amine works as a protection group to move the reactive center towards the acid functionality of the L-Phenylalanine. Besides monitoring the progress of the reaction, the NMR spectra also provide information about the addition of reactants step and the consequent hydrolysis of the reactant acetic anhydride. Read more …
Figure. Left: NMR spectra of reactant (bottom) and product (top) measured in a 5 mm NMR tube. Right: Stacked plot showing the spectra (decimated by factor of 4) collected during a time of 160 min. Each spectrum was acquired using two scans collected with a repetition time of 30 seconds. Integrals plotted over time of unprotected (blue) and protected (red) L-Phenylalanine.
Fluorine containing scaffolds and building blocks play a remarkable role within drug discovery in medicinal chemistry for the synthesis of Active Pharmaceutical Ingredients (API). The fluorine atom holds some key advantages in terms of pharmacokinetic and physicochemical properties. Some of the major advantages are the improved metabolic stability and the enhanced membrane permeation that allow for a significantly higher bioavailability of fluorine-containing drugs. On-line reaction monitoring by NMR has proven to be a valuable tool in process control, as it allows for a deep insight into mechanisms and kinetics of chemical reactions at laboratory, as well as pilot-scale. In previous Application Notes, we have demonstrated the benefits of using the Spinsolve Reaction Monitoring Kit to follow the progress of different reactions by 1H-NMR spectroscopy. In this application note, we show how the course of an SNAr reaction can be monitored on-line by acquiring both 1H- and 19F-NMR spectra sequentially in a continuous mode. For these particular experiments we employed our Spinsolve 60 MHz HF ULTRA system, but all Spinsolve models come with capabilities to acquire 1Hand 19F-NMR spectra without retuning or recalibration. Read more …
Figure. Left: SNAr reaction for the synthesis of 4-(2-Fluoro-4-nitrophenyl)morpholine (3) with 1H-NMR spectra (stacked) of the starting materials and the reaction mixture all recorded separately. Right: 1H-NMR spectra (stacked left side) and 19F-NMR spectra (stacked right side) of the kinetic study with an overall spectra number of 124 for each nucleus recorded sequentially. Trends of 1H NMR spectra over reaction time based on integrated regions of interest for reactants morpholine (2) and 1,2-Difluoro-4-nitrobenzene (1) and three product 3 specific ranges.
The on-line monitoring of chemical reactions employing NMR spectroscopy provides a deeper insight into the kinetics of the reaction in question and its mechanism. Several nuclei like 1H, 19F or 31P can be detected exclusively or combined in a consecutive experiment loop to follow the course of the reaction and to identify and quantify the chemical moieties of choice as well as possible intermediates or side-products. Within stereoselective synthesis, besides the conversion of the starting material the decisive step for the operator is to define the major and minor stereoisomers of the desired product and to optimize the chemical process towards complete stereoselectivity. For the quantification of the stereoselective outcome of chemical reactions e.g. the determination of the enantiomeric excess of the reaction product, typically HPLC methods with chiral columns and reference compounds are considered. Nevertheless, when dealing with diastereoisomers as possible reaction products the quantification of the diastereomeric ratio can be determined directly by NMR spectroscopy without the need for reference compounds or a calibration. Within the following application note, we demonstrate for the first time how benchtop NMR spectroscopy can be employed to not only follow the course of a stereoselective reaction on-line but also to determine its stereochemical outcome. For this purpose, the well-known Horner-Wadsworth-Emmons reaction (HWE reaction) was selected using our latest member of the Spinsolve family the Spinsolve 80 MHz ULTRA system. Read more …
Figure. Left: 1H-NMR spectra (stacked) of the starting materials recorded separately as well as the reaction mixture (last spectrum of 500 shown). Right: Last acquired 1H-NMR spectra (stacked) of reactions with and without TMEDA. 1H-NMR spectra (stacked plot at the bottom; due to visibility reasons a decimation of two was applied) of the kinetic study with an overall spectra number of 500 including the trends of the integrals over time of the regions of interest.
The development of high-performance benchtop NMR spectrometers provides a practical and information-rich way to monitor on-line/in-line the progress of chemical reactions. NMR provides not only the structural information about the different chemical species involved in the reaction but also quantitative information about the concentration of reactants and products. By following the conversion in in real time, chemists get the required insight to understand the kinetics of the reaction. Another advantage of using NMR for reaction monitoring is the ability to observe reaction intermediates. These can often be missed with endpoint reaction analysis, since intermediate, by definition, gets produced in the early stage and consumed during a later stage of the reaction. In recent years, the availability and affordability of benchtop NMR systems have offered the opportunity for schools to incorporate NMR spectroscopic techniques into organic chemistry curricula. With simple button-clicking, instructors and students can perform different NMR experiments to confirm the reaction products, or to monitor a reaction with NMR. This application note describes an example of using the Spinsolve 60 MHz Carbon ULTRA to monitor a frequently used reaction in the organic chemistry laboratory – the Claisen-Schmidt (or cross-aldol) condensation reaction to synthesize dibenzalacetone. Students can visualize the kinetic profile of different chemical components in real-time. They can also observe the reaction intermediate, which is a concept that is seldomly demonstrated in laboratory experiments since intermediate species are not easily isolated from the reaction mixture for characterization. With the hands-on experience in the lab, students will develop a deeper understanding of chemistry concepts that they learn in lectures and familiarize themselves with the modern NMR techniques being adopted in industry. Read More…
Figure. Left: Claisen-Schmidt condensation reaction for the synthesis of dibenzalacetone analog. Right: Stack plot of 1H NMR spectra of the reaction progress and integral over time plot of the defined integral regions.
Further Reading
Publications:
Alexander P. Grimm, Stephen T. Knox, Clarissa Y. P. Wilding, Harry A. Jones, Björn Schmidt, Olga Piskljonow, Dominik Voll, Christian W. Schmitt, Nicholas J. Warren, Patrick Théato, MacromolecularRapidCommunications, (2025)
Real-Time Monitoring of Fermentation Processes in Wine Production with Benchtop 1H NMR Spectroscopy
Johnnie Phuong, Billy Salgado, Jana Heiß, Ellen Steimers, Patrick Nickolaus, Lena Keller, Ulrich Fischer, Erik von Harbou, Daniel J. Holland, Fabian Jirasek, Hans Hasse, Kerstin Münnemann, JFoodResearchInt, (2025)
Automated self-optimization, intensification, and scale-up of photocatalysis in flow
Aidan Slattery, Zhenghui Wen, Pauline Tenblad, Jesús Sanjosé-Orduna, Diego Pintossi, Tim den Hartog, Timothy Noël, Science, (2024)
Tristan Maschmeyer, David J. Russell, José G. Napolitano and Jason E. Hein, Magnetic Resonance in Chemistry (2023)
Reaction Kinetics using a Chemputable Framework for Data Collection and Analysis
Bartosz M. Matysiak, Dean Thomas and Leroy Cronin, Angewandte Chemie International Edition (2023)
Control of a complex multistep process for the production of mesalazine
Ismael Castillo, Jakob Rehrl, Peter Sagmeister, René Lebl, Julia Kruisz, Selma Celikovic, Martin Sipek, Dirk Kirschneck, Martin Horn, Stephan Sacher, David Cantillo, Jason D. Williams, Johannes G. Khinast and C. Oliver Kappe; Journal of Process Control (2023)
Advanced Real-Time Process Analytics for Multistep Synthesis in Continuous Flow
Peter Sagmeister, René Lebl, Ismael Castillo, Jakob Rehrl, Julia Kruisz, Martin Sipek, Martin Horn, Stephan Sacher, David Cantillo, Jason D. Williams, C. Oliver Kappe; Angewandte Chemie International Edition; (2021)
Dylan Bouillaud, Delphine Drouin, Benoît Charrier, Corentin Jacquemmoz, Jonathan Farjon, Patrick Giraudeau, Olivier Gonçalves, Process Biochemistry, (2020)
Online process monitoring of a batch distillation by medium field NMR spectroscopy
Anne Friebel, Erik von Harbou, Kerstin Münnemann, Hans Hasse, G., Chem. Eng. Science, (2020)
An Artificial Intelligence that Discovers Unpredictable Chemical Reactions
Dario Caramelli, Jarosław M. Granda, Dario Cambié, S. Hessam M. Mehr, Alon Henson and Leroy Cronin, ChemRxiv, (2020)
Controlling an organic synthesis robot with machine learning to search for new reactivity
Jarosław M. Granda, Liva Donina, Vincenza Dragone, De-Liang Long and Leroy Cronin, Nature, (2018)
Simon Kern, Klas Meyer, Svetlana Guhl, Patrick Gräßer, Andrea Paul, Rudibert King, Michael Maiwald, Anal. Bioanal. Chem., (2018)
Benchtop NMR for online reaction monitoring of the biocatalytic synthesis of aromatic amino alcohols
Christiane Claaßen, Kevin Mack, Dörte Rother, ChemCatChem, (2019)
Stephen T. Knox, Sam Parkinson , Raphael Stone and Nicholas J. Warren, Polymer Chemistry, (2019
Download Resources
- Brochure Reaction Monitoring
- App Note Simultaneous Dual-Phase Reaction Monitoring by Benchtop NMR
- App Note Automated optimization of a flow reactor by using Bayesian algorithms and Inline NMR Monitoring
- App Note Monitoring the oxidation of Phosphine ligands using 31P NMR
- App Note Transesterification of Pinacol Arylboronic Ester – How to monitor heterogeneous reactions online by benchtop NMR
- App Note Monitoring the deuteration of cinnamyl alcohol in a flow reactor with a Spinsolve Benchtop NMR
- App Note Optimizing a two-step hydrogenation reaction in a flow reactor using a Spinsolve Benchtop NMR
- App Note Coupling a Spinsolve® NMR spectrometer to an H-Cube® Pro flow reactor for fast reaction optimization
- App Note Monitoring a Claisen-Schmidt Condensation Reaction by NMR in the teaching Class
- App Note Quantifying the formation of stereoisomers by benchtop NMR spectroscopy
- App Note Online monitoring of the formation of a diimine
- App Note Online Monitoring of the N-acetylation Reaction of L-Phenylalanine
- App Note On-line reaction monitoring of an SNAr reaction by 1H and 19F NMR
- App Note Benchtop NMR Reaction Monitoring for Kinetic Profiling
- Video Reaction Monitoring Software Demo